Abstract
Introduction
Heparin Induced Thrombocytopenia (HIT) is a severe adverse reaction to heparin treatment characterized by antibodies to platelet factor 4 (PF4)-heparin complexes. Recent data suggests that currently used therapies are still associated with significant incidence of thrombosis, amputation and death (Kuter et al, AJH 2017 Aug; 92(8):730-73). Thus, several off-label treatments such as therapeutic plasma exchange (TPE; Blood . 2015 Jan 1;125(1):195-8) and IVIg (Padmanabhan et al, Chest. 2017 Apr 17 epub) are increasingly being used to treat severe or refractory cases. In performing TPE, a portion of the patient's plasma is removed and substituted by a replacement fluid, typically 5% human albumin and less frequently, normal plasma. Our recent work (Padmanabhan et al, Chest. 2017 Apr 17 epub extending Rollin et al, Blood 2015) demonstrates that normal immunoglobulin G is a potent inhibitor of HIT antibody-mediated platelet activation and that platelets from patients with the 131HH genotype and to a lesser extent 131RR/HR genotypes of the platelet IgG receptor, FcγRIIa, are susceptible to this inhibition. TPE impacts plasma levels of immunoglobulin in two ways -- it decreases the titer of pathogenic HIT antibodies and it depletes non-pathogenic plasma IgGs. The latter effect could theoretically counteract the salutary impact of HIT antibody removal by TPE and potentially even worsen disease. This study was carried out to assess the impact of the diluent used for exchange on HIT-antibody mediated platelet activation, using an in vitro model of TPE.
Methods
Samples from two patients with severe HIT requiring TPE treatment were used for these studies. The PF4-dependent p-selectin expression assay (PEA) using platelets treated with low-dose PF4 (1.25mcg/ml) was performed as described (Padmanabhan et al, Chest. 2017 Apr 17 epub) using two blood group O platelet donors, possessing FcγRIIa genotype 131HH and 131RR, respectively. A one-volume plasma exchange removes about 60% of an intravascular substance assuming no recirculation or rapid intravascular-extravascular exchange. To mimic TPE in an in vitro system , HIT samples were diluted 1:2 or 1:4 in normal plasma, IgG-depleted normal plasma, or IgG depleted normal plasma to which IVIg had been added to attain normal IgG levels ("IgG-repleted plasma") and then tested in triplicate in the PEA. A p-value of <0.05 was considered significant in the student's ttest (<0.05[*], <0.01 [**], <0.001 [***], <0.0001 [****])
Results
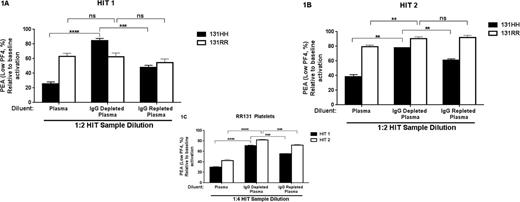
We observed a significant decrease in p-selectin expression with both HIT antibodies to ~25% and ~40% of baseline when 131HH platelets were used for testing and the HIT samples were diluted in normal plasma (Figs 1A, B, closed bars, left panel). PEA results rose sharply to ~85% and ~80% of baseline when IgG-depleted normal plasma was used in place of unmanipulated normal plasma (Figs 1A, B, closed bars, middle panel). Use of IgG repleted plasma decreased PEA reactions to ~50% and ~60% of baseline (Figs 1A, B, closed bars, right panel). As expected, when 131RR platelets were used, IgG depletion/repletion status of the normal plasma had little/no effect on PEA results (Figs 1A, B, open bars), however, when a higher dilution of HIT serum (1:4) was used to mimic serial TPE (Fig 1C), IgG replete solutions had maximal inhibitory effect on platelet activation.
Conclusion
TPE, typically performed with 5% albumin replacement, is an important therapeutic modality in patients with acute/subacute HIT who require heparin exposure (e.g. during emergency cardiac surgery) and in severe HIT refractory to standard treatment (e.g. worsening thrombosis). Studies demonstrating IVIg-mediated inhibition of HIT antibody-induced platelet activation suggested that normal plasma (IgG replete) should be more effective than albumin in maintaining platelet quiescence. Results obtained in our in vitro TPE system show that plasma depleted of IgG is much less effective in inhibiting platelet activation by HIT antibodies, whereas plasma with intact IgG is highly effective. While minimal/no effect of IgG on antibody-induced activation of 131RR platelets was noted at the dilution mimicking one TPE treatment (1:2 dilution), a strong inhibitory effect was seen at higher dilutions (as would be seen in patients undergoing serial TPE). These results suggest that plasma (or IVIg infusion following albumin-based TPE) is the preferred replacement solution for TPE in HIT.
Jones: BloodCenter of Wisconsin: Patents & Royalties: A patent application has been filed related to the PF4-dependent P-selectin expression assay (Method of Detecting Platelet- Activating Antibodies That Cause Heparin-Induced Thrombocytopenia/Thrombosis; PCT/US14/62591).. Curtis: Ionis Pharmaceuticals: Consultancy. Bougie: BloodCenter of Wisconsin: Patents & Royalties: A patent application has been filed related to the PF4-dependent P-selectin expression assay (Method of Detecting Platelet- Activating Antibodies That Cause Heparin-Induced Thrombocytopenia/Thrombosis; PCT/US14/62591).. Aster: BloodCenter of Wisconsin: Patents & Royalties: A patent application has been filed related to the PF4-dependent P-selectin expression assay (Method of Detecting Platelet- Activating Antibodies That Cause Heparin-Induced Thrombocytopenia/Thrombosis; PCT/US14/62591)., Research Funding. Padmanabhan: Retham Technologies LLC: Equity Ownership; BloodCenter of Wisconsin: Patents & Royalties: A patent application has been filed related to the PF4-dependent P-selectin expression assay (Method of Detecting Platelet- Activating Antibodies That Cause Heparin-Induced Thrombocytopenia/Thrombosis; PCT/US14/62591).
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal